Vietnam becomes 1st country to authorise commercial circulation of African swine fever vaccine
NDO/VNA - Vietnam has become the first country in the world to successfully research and produce a vaccine
Deputy Minister of Agriculture and Rural Development Phung Duc Tien informed about the results of research and production of the African swine fever vaccine. Photo: Tung Dinh.
The African Swine Fever (ASF) first appeared in Vietnam in February 2019 and quickly spread across the country, forcing the destruction of approximately 6 million pigs, resulting in a loss of VND 30 trillion and having a direct impact on the CPI index of 2020.
Currently, the outbreak continues in several locations around the nation. Without an effective vaccine, the risk of ASF continuing to emerge and spread is very high due to the virus's ability to survive in the environment for an extended period and the transmission's complexity; meanwhile, household farming still accounts for a large proportion of livestock production and has not yet met biosecurity requirements.
Since the emergence of African swine fever (ASF) in Vietnam, all levels of government have intervened to aggressively direct and synchronize measures to prevent and battle the disease, as well as to coordinate the development of a preventative vaccine.
Ministry of Agriculture and Rural Development also instructed the Department of Animal Health (DAH) and businesses with the ability, strength, and expertise to conduct ASF vaccine research, production, and registration.
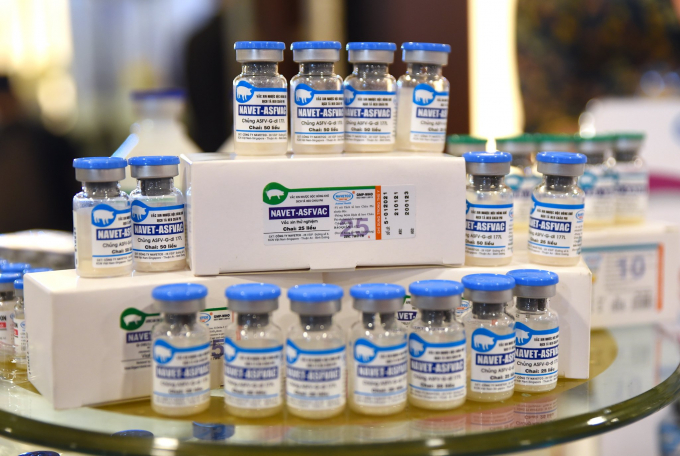
Navetco's NAVET-ASFVAC African swine fever vaccine will foster the livestock industry in coping with the disease more effectively. Photo: Tung Dinh.
As soon as American researchers announced the successful removal of the ASF-G-Delta I177L gene from an attenuated ASF virus strain at the beginning of November 2019, the leaders of the MARD sent leaders of the DAH to the United States to attend meetings and discuss directly with US experts the coordination of research and production of ASF vaccines.
As of February 2020, Vietnamese vaccine research and production institutes officially coordinated with the US experts. The Ministry instructed DAH to finalize and sign the Memorandum of Understanding of technical cooperation with the Agricultural Research Service of the United States Department of Agriculture to provide the framework for the execution of the missions.
The Ministry has approved the importation of gene-deleted attenuated ASF virus strains for research and manufacture of ASF vaccines in Vietnam beginning in July 2020.
As soon as Navetco Company received the ASFV-G-I177L strain from the United States Department of Agriculture in September 2020, it immediately started research, conducting five laboratory experiments.
100% of vaccinated pigs were protected against the virus under laboratory circumstances, whereas 80% of vaccinated pigs were protected against the viral strain in Vietnam under production conditions. The duration of the vaccine's protection is six months after immunization.
NAVET-ASFVAC African swine fever vaccine of Navetco Company.
MARD established scientific councils at the grassroots and ministerial levels and convened dozens of meetings with the participation of many leading Vietnamese scientists, managers, and manufacturers of veterinary vaccines to investigate, and evaluate very carefully the report on scientific research results and the application for registration of circulation of the ASF NAVET-ASFVAC vaccine developed by Navetco.
The research and production findings of NAVET - ASFVAC of Navetco have been accepted and published in the Journal of Veterinary Science in Vietnam and other prestigious scientific publications across the world.
The Agricultural Research Service of the United States Department of Agriculture confirmed the safety and efficacy of the NAVET-ASFVAC vaccination in an official document sent to the Department of Animal Health of Vietnam on May 17, 2022.
Vietnam becomes the first nation in the world to manufacture a vaccine against this severe illness in pigs, proving the effective collaboration efforts of the veterinary industry, enterprises, and Vietnamese scientists with global scientists to master vaccination research and production technologies. This, as underlined by Deputy Minister Phung Duc Tien, is a crucial premise for disease prevention in cattle herds.
In the future, it is anticipated that MARD will direct the organization of research and evaluation of the quality of vaccines for other pig objectives (sows, boars); duration of immunity, vaccine stability under different storage and use conditions; and evaluation of risk factors that may affect the quality and effectiveness of vaccines in production and livestock conditions.
ASF is not transmitted to humans, however, it has a high mortality rate among pigs of all ages and species (domestic pigs and wild boars).
The ASF virus is very resistant to environmental conditions and stays infectious for extended periods of storage. The infected pig may become the virus's permanent host.
Even though almost 4,000 scientific studies on the ASF virus and vaccine development have been published, no commercial vaccine against ASF has been announced.
According to data from the World Organization for Animal Health (OIE) and the Food and Agriculture Organization of the United Nations (FAO), ASF is a threat to the global swine industry.
On the basis of reports on the careful research, evaluation, and appraisal of the Department of Health; concluding comments and suggestions of Ministerial-level Scientific Councils for registration of circulation of veterinary vaccines; and the conclusion that the application for registration for circulation of the NAVET-ASFVAC vaccine of Navetco Company fully satisfies the requirements for issuance of a Certificate of Sale in accordance with the provisions of the Law on Animal Health, the guiding documents of the Law. MARD directs the Department of Animal Health to assume the prime responsibility for, and coordinate with relevant agencies in granting the Certificate of Circulation for the Navetco Company's NAVET-ASFVAC vaccine according to regulations.
Additionally, the sterility, safety, and efficacy of 10 consecutive batches of NAVET-ASFVAC vaccination must be rigorously monitored.
Monitoring the use of the NAVET-ASFVAC vaccine following its commercialization in two phases:
Phase 1: Use the NAVET-ASFVAC vaccine in a limited area; 600,000 doses of the vaccine are anticipated to be available for purchase.
Phase 2: Once the report on the evaluation of the outcomes of administering the NAVET-ASFVAC vaccine in phase 1 is ready, the Department of Animal Health submits a report to the Ministry of Agriculture and Rural Development to direct the nationwide use of the vaccine.
Translated by Linh Linh